Armed with $3.25 million CAD in seed funding, Montréal-based healthtech startup Ditch Labs wants to help smokers wean themselves off nicotine with its dual hardware-software solution.
The early-stage startup has set its sights on a massive and increasing public health issue: nicotine addiction. A highly-addictive stimulant found in tobacco products, nicotine is why so many smokers find it tough to quit using cigarettes and vaporizers.
“Current solutions don’t work. They usually try to treat a very complex problem … with a very one-size-fits-all approach.”
– Laurent Laferrière, Ditch Labs
Smoking represents one of the leading causes globally of preventable disease, disability, and death. Each year, about 48,000 Canadians die from tobacco use, per the Canadian Centre on Substance Use and Addiction. According to a recent Health Canada survey, the problem is growing: Canada boasts some of the highest teen vaping rates in the world.
From the lives lost, to the associated healthcare costs of smoking, “The scale of the problem is huge,” Ditch Labs co-founder and CEO Laurent Laferrière told BetaKit in an interview.
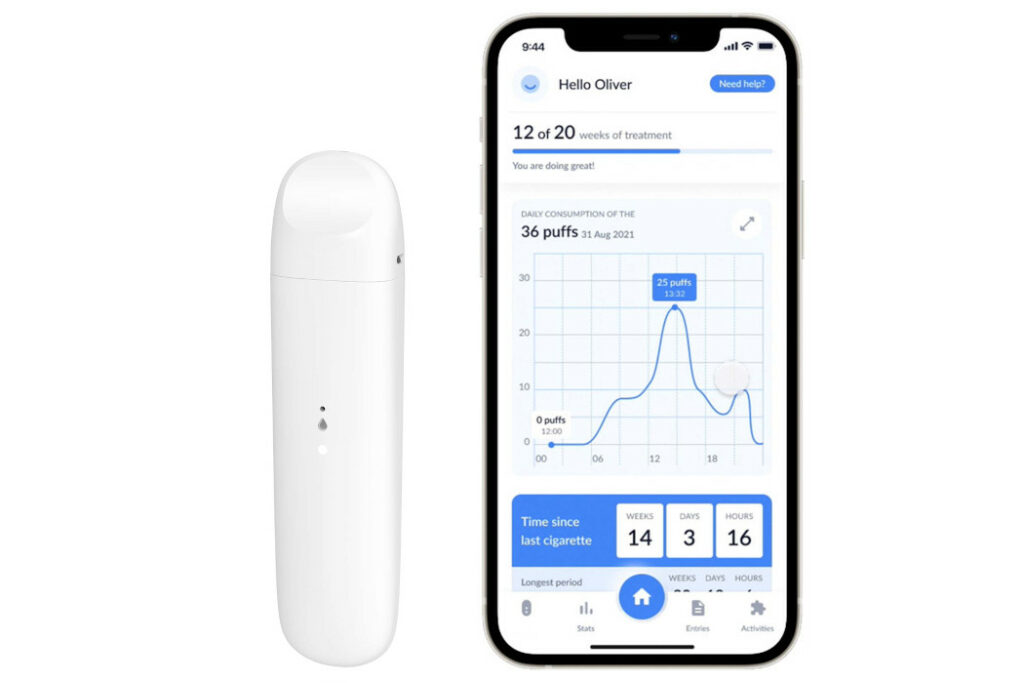
Founded in 2020 by Laferrière, CTO Olivier Bourbonnais, and chief scientific officer Christelle Luce—who holds a PhD in psychology with a focus on addiction—Ditch Labs is developing a smoking cessation treatment for nicotine addiction that involves a proprietary medical device paired with a therapeutic mobile application.
Ditch Labs’ solution consists of a medical nicotine vaporizer that facilitates controlled delivery and a complementary app that provides personalized support, progress tracking, and behavioural interventions. With this combo, the startup aims to address the physiological and psychological impacts of nicotine addiction simultaneously.
The company’s convertible note seed financing closed in March and was led by Amplify Capital and Boreal Ventures, with support from fellow existing backers Symphony Health Network and Anges Québec. The round also saw participation from new investors Investissement Québec, Meditrial, Kale Fund, and nine undisclosed doctors from the United States and Canada. This new capital brings Ditch Labs’ total funding to $5.7 million.
A variety of over-the-counter and prescription solutions designed to help people quit smoking exist, from nicotine patches and gums to therapy.
According to the United States (US) Centers for Disease Control and Prevention (CDC), a majority of smokers want to quit and have tried to do so. However, the CDC reports that only a small portion actually succeed.
“Current solutions don’t work,” argued Laferrière. “They usually try to treat a very complex problem … with a very one-size-fits-all approach.”
According to the CEO, Ditch Labs decided to take a “more holistic” approach that currently offers 40 different treatments for nicotine addiction. The firm’s solution also leverages a vaping device, which has become a popular tool among smokers looking to stop. Ditch Labs’ hardware is designed to control doses and reduce them automatically over time. Laferrière added that Ditch Labs has mechanisms in place to prevent over-use, including the ability to deploy placebos once users hit their daily limit.
RELATED: Cannabis vaporizer startup Greentank Technologies closes $22.2 million CAD Series B
Laferrière acknowledged that there are also other tech startups trying to help tackle this problem, from firms building standalone apps to players that have taken a similar hardware-software approach to Ditch Labs.
Compared to firms in the latter group, the CEO argued that Ditch Labs’ biggest advantage is its intellectual property. Per Laferrière, Ditch Labs holds patents in 15 countries for “dual-tank, dual-coil technology.” This two-tank, two-system vaporizer tech enables it to vary doses and distribute placebos to users.
Laferrière said Ditch Labs has already had several discussions with Health Canada and the US Food and Drug Administration. According to the CEO, given the complexity of the problem Ditch Labs aims to solve, and the regulatory requirements it must satisfy, the startup remains roughly three years away from being able to commercialize its tech in Canada and the US.
The startup’s next step will be obtaining clinical data concerning the efficacy of its approach, which it hopes to gather through its upcoming, 350-person clinical trial, which Ditch Labs plans to launch this fall.
Feature image courtesy Ditch Labs.


